|

Almost
any human cell can be used to replicate a person via cloning
CRISPR is a technology that can be used to edit genes and, as such, will likely change the world.
The essence of CRISPR is simple: it’s a way of finding a specific bit of DNA inside a cell. After that, the next step in CRISPR gene editing is usually to alter that piece of DNA. However, CRISPR has also been adapted to do other things too, such as turning genes on or off without altering their sequence.
There were ways to edit the genomes of some plants and animals before the CRISPR method was unveiled in 2012 but it took years and cost hundreds of thousands of dollars. CRISPR has made it cheap and easy.
CRISPR is already widely used for scientific research, and in the not too distant future many of the plants and animals in our farms, gardens or homes may have been altered with CRISPR. In fact, some people already are eating CRISPRed food.
CRISPR technology also has the potential to transform medicine, enabling us to not only treat but also prevent many diseases. We may even decide to use it to change the genomes of our children. An attempt to do this in China has been condemned as premature and unethical, but some think it could benefit children in the future.
CRISPR is being used for all kinds of other purposes too, from fingerprinting cells and logging what happens inside them to directing evolution and creating gene drives.
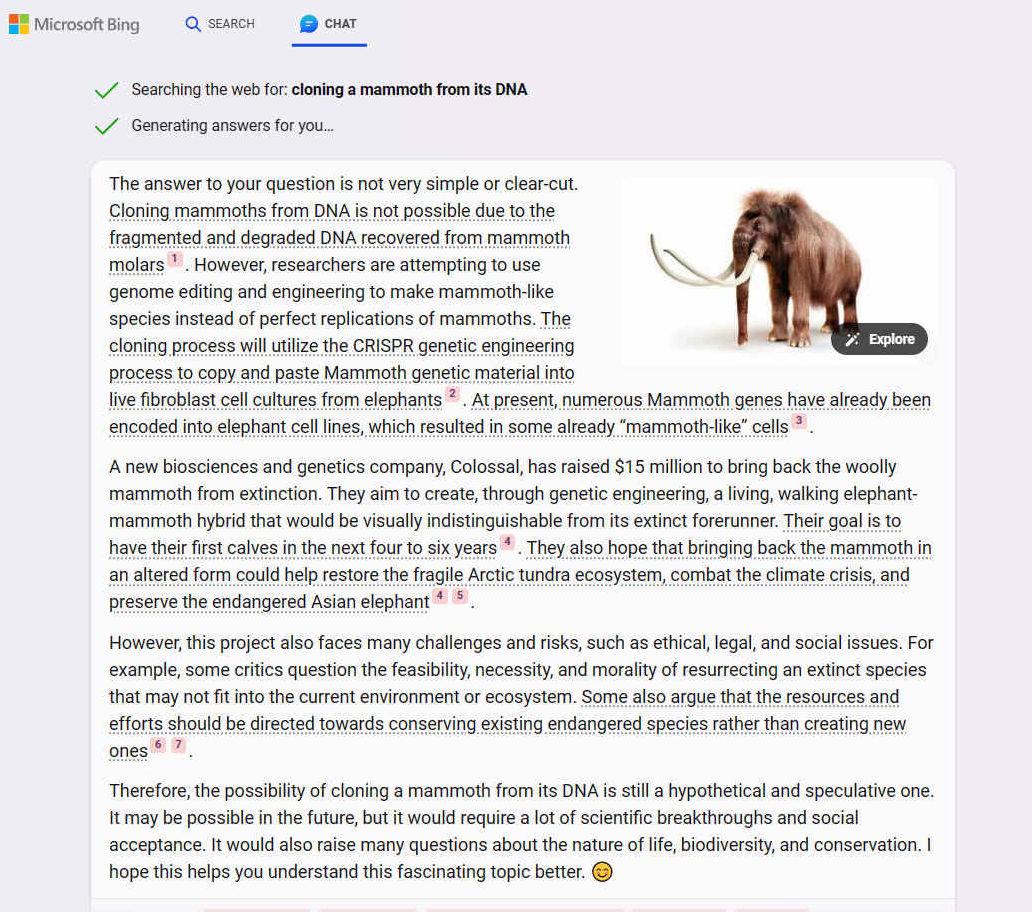
The key to CRISPR is the many flavours of “Cas” proteins found in bacteria, where they help defend against viruses. The Cas9 protein is the most widely used by scientists. This protein can easily be programmed to find and bind to almost any desired target sequence, simply by giving it a piece of RNA to guide it in its search.
When the CRISPR Cas9 protein is added to a cell along with a piece of guide RNA, the Cas9 protein hooks up with the guide RNA and then moves along the strands of DNA until it finds and binds to a 20-DNA-letter long sequence that matches part of the guide RNA sequence. That’s impressive, given that the DNA packed into each of our cells has six billion letters and is two metres long.
This
means significant
computing power is needed when working with DNA subject
identification and analysis.
What happens next can vary. The standard Cas9 protein cuts the DNA at the target. When the cut is repaired, mutations are introduced that usually disable a gene. This is by far the most common use of CRISPR. It’s called genome editing – or gene editing – but usually the results are not as precise as that term implies.
CRISPR can also be used to make precise changes such as replacing faulty genes – true genome editing – but this is far more difficult.
Customised Cas proteins have been created that do not cut DNA or alter it in any way, but merely turn genes on or off: CRISPRa and CRISPRi respectively. Yet others, called base editors, change one letter of the
DNA code to another.
So why do we call it CRISPR? Cas proteins are used by bacteria to destroy viral DNA. They add bits of viral DNA to their own genome to guide the Cas proteins, and the odd patterns of these bits of DNA are what gave CRISPR its name: clustered regularly interspaced short palindromic repeats.
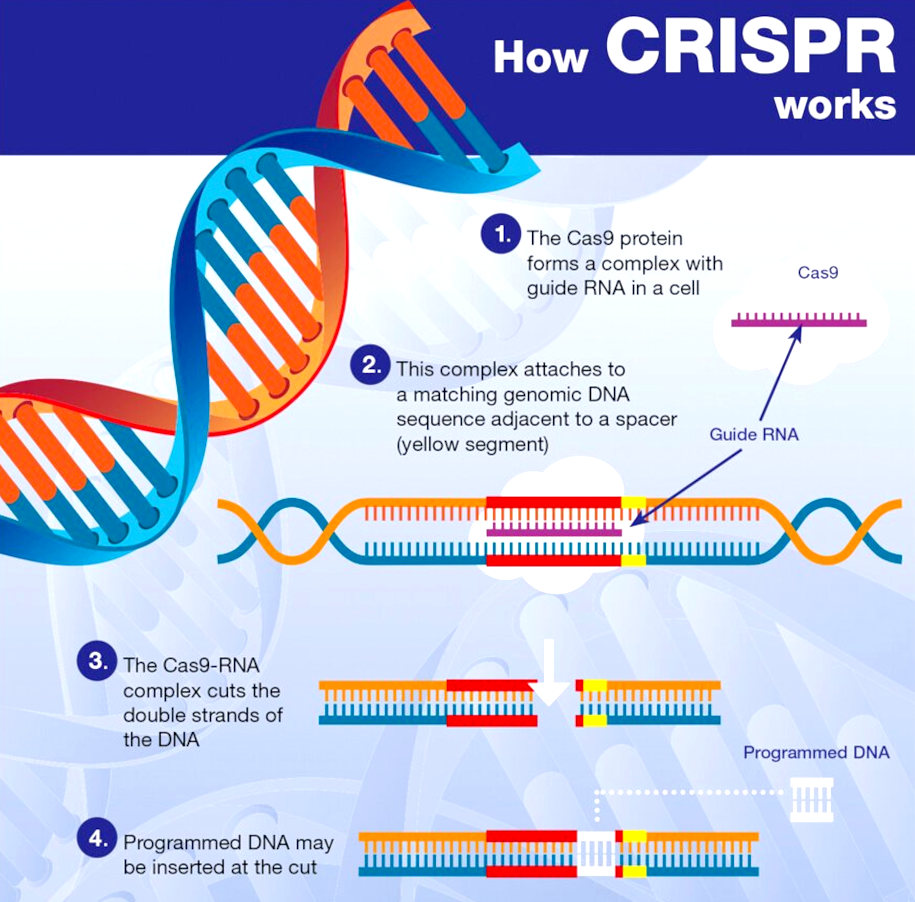
CRISPR gene editing is a
genetic engineering technique in molecular biology by which the genomes of living organisms may be modified. It is based on a simplified version of the bacterial CRISPR-Cas9 antiviral defense system. By delivering the Cas9 nuclease complexed with a synthetic guide RNA (gRNA) into a cell, the cell's genome can be cut at a desired location, allowing existing genes to be removed and/or new ones added in vivo.
The technique is considered highly significant in biotechnology and medicine as it allows for the genomes to be edited in vivo with extremely high precision, cheaply, and with ease. It can be used in the creation of new medicines, agricultural products, and genetically modified organisms, or as a means of controlling pathogens and pests. It also has possibilities in the treatment of inherited genetic diseases as well as diseases arising from somatic mutations such as cancer. However, its use in
human germline genetic modification is highly controversial. The development of the technique earned Jennifer Doudna and Emmanuelle Charpentier the Nobel Prize in Chemistry in 2020. The third researcher group that shared the Kavli Prize for the same discovery, led by Virginijus Šikšnys, was not awarded the Nobel prize.
Working like genetic scissors, the Cas9 nuclease opens both strands of the targeted sequence of DNA to introduce the modification by one of two methods. Knock-in mutations, facilitated via homology directed repair (HDR), is the traditional pathway of targeted genomic editing approaches. This allows for the introduction of targeted DNA damage and
repair, such as may be needed in cloning
from ancient mummy DNA.
Or mammoth replication. HDR employs the use of similar DNA sequences to drive the repair of the break via the incorporation of exogenous DNA to function as the repair template. This method relies on the periodic and isolated occurrence of
DNA damage at the target site in order for the repair to commence. Knock-out mutations caused by CRISPR-Cas9 result in the repair of the double-stranded break by means of non-homologous end joining (NHEJ). NHEJ can often result in random deletions or insertions at the repair site, which may disrupt or alter gene functionality. Therefore, genomic engineering by CRISPR-Cas9 gives researchers the ability to generate targeted random gene disruption. Because of this, the precision of genome editing is a great concern. Genomic editing
can lead to potentially irreversible changes to the genome.
BIOMEDICINE
CRISPR-Cas technology has been proposed as a treatment for multiple human diseases, especially those with a genetic cause. Its ability to modify specific DNA sequences makes it a tool with potential to fix disease-causing mutations. Early research in animal models suggest that therapies based on CRISPR technology have potential to treat a wide range of diseases, including cancer, progeria, beta-thalassemia, sickle cell disease, hemophilia, cystic fibrosis, Duchenne's muscular dystrophy, Huntington's disease, transthyretin amyloidosis and heart disease. CRISPR has also been used to cure malaria in mosquitos, which could eliminate the vector and the disease in humans. CRISPR may also have applications in tissue engineering and regenerative medicine, such as by creating human blood vessels that lack expression of MHC class II proteins, which often cause transplant rejection.
In addition, clinical trials to cure beta thalassemia and sickle cell disease in human patients using CRISPR-Cas9 technology have shown promising results.
POLICIES
Policy regulations for the CRISPR-Cas9 system vary around the globe. In February 2016, British scientists were given permission by regulators to genetically modify
human embryos by using CRISPR-Cas9 and related techniques. However, researchers were forbidden from implanting the embryos and the embryos were to be destroyed after seven days.
The US has an elaborate, interdepartmental regulatory system to evaluate new genetically modified foods and crops. For example, the Agriculture Risk Protection Act of 2000 gives the United States Department of
Agriculture the authority to oversee the detection, control, eradication, suppression, prevention, or retardation of the spread of plant pests or noxious weeds to protect the agriculture, environment, and economy of the US. The act regulates any genetically modified organism that utilizes the genome of a predefined "plant pest" or any plant not previously categorized. In 2015, Yinong Yang successfully deactivated 16 specific genes in the white button mushroom to make them non-browning. Since he had not added any foreign-species (transgenic) DNA to his organism, the mushroom could not be regulated by the USDA under Section 340.2. Yang's white button mushroom was the first organism genetically modified with the CRISPR-Cas9 protein system to pass US regulation.
In 2016, the USDA sponsored a committee to consider future regulatory policy for upcoming genetic modification techniques. With the help of the US National Academies of Sciences, Engineering, and Medicine, special interests groups met on April 15 to contemplate the possible advancements in genetic engineering within the next five years and any new regulations that might be needed as a result. In 2017, the
Food and Drug Administration proposed a rule that would classify genetic engineering modifications to animals as "animal drugs", subjecting them to strict regulation if offered for sale and reducing the ability for individuals and small businesses to make them profitable.
In China, where social conditions sharply contrast with those of the West, genetic diseases carry a heavy stigma. This leaves China with fewer policy barriers to the use of this technology.
What is CRISPR genetic engineering?
- CRISPR genetic engineering is a technique that allows scientists to modify the DNA of living organisms with high precision, speed, and ease. CRISPR stands for Clustered Regularly Interspaced Short Palindromic Repeats, which are sequences of DNA that are found in bacteria and other microorganisms. CRISPR is part of a natural defense system that bacteria use to fight off viruses by cutting and destroying their DNA. Scientists have adapted this system to create a tool that can cut and edit any DNA sequence in any organism by using a protein called Cas9 and a guide RNA that matches the target DNA. CRISPR genetic engineering has many potential applications in biotechnology, medicine, and agriculture, such as creating new medicines, improving crops, or curing diseases. However, CRISPR genetic engineering also raises ethical, legal, and social issues, such as the safety, regulation, and morality of altering the genomes of living beings [1] [2] [3]. I hope this helps you understand what CRISPR genetic engineering is. ��
CRISPR
IN FICTION
In
the John
Storm adventure series, German, Italian and Swiss
scientists Rudolph
Kessler, Franco
Francisco and Klaus
von Kolreuter, have developed a method of gene splicing
using supercomputers and a DNA database they have been
adding to by stealing from Scotland
Yard's criminal facility and other medical
establishments. Giving blood to your doctor for routine
tests, is one way National Health Services accumulate data,
that is passed upstream, despite data subject protection
laws. In addition to genetically engineering while
cloning and replicating humans, they have also perfected a
microbiological serum at molecular level using CRISPR
technology, to be able to super enhance subjects in vitro.
Though, the Panamanian
Running Man, was engineered at DNA level, before being
replicated.
REFERENCE
https://en.wikipedia.org/wiki/CRISPR_gene_editing
https://bing.com/search?q=CRISPR+genetic+engineering
https://sitn.hms.harvard.edu/flash/2014/crispr-a-game-changing-genetic-engineering-technique/
https://www.newscientist.com/definition/what-is-crispr/
https://www.addgene.org/guides/crispr/
https://en.wikipedia.org/wiki/CRISPR_gene_editing
https://bing.com/search?q=CRISPR+genetic+engineering
https://sitn.hms.harvard.edu/flash/2014/crispr-a-game-changing-genetic-engineering-technique/
https://www.newscientist.com/definition/what-is-crispr/
https://www.addgene.org/guides/crispr/

CLEOPATRA
THE MUMMY - UNDER DEVELOPMENT
'Cleopatra
- The Mummy' is the proposed sequel to 'Kulo-Luna.'
Kulo-Luna, the first script of the John Storm franchise (for which a draft
is available to studios and actor's agents). The John Storm
franchise is a series of
ocean awareness adventures, featuring the incredible solar
powered trimaran: Elizabeth
Swann. 'Cleopatra The Mummy,' could be the pilot, with
Kulo-Luna, or Treasure Island the prequel or sequel. The
order of production could be to suit identified gaps in
entertainment, in any particular year. Equally, the trilogy,
could be adapted for network television, as with Blood
and Treasure from CBS.
|








